No Products in the Cart
When we talk about liquid oxygen, the first number you need to know is -183°C (-297°F). This isn't just some abstract figure; it's the boiling point, the critical threshold where liquid oxygen decides it would rather be a gas. Everything about handling, storing, and using this substance revolves around staying on the cold side of that number.
Think about boiling a kettle. At 100°C, water turns into steam. Liquid oxygen works on the same principle, just at the extreme opposite end of the thermometer. That -183°C mark is its boiling point, and if the temperature creeps even a fraction of a degree above that, it starts boiling off vigorously.
This intense cold is what makes liquid oxygen, or sauerstoff flüssig temperatur as it's known in technical German contexts, so incredibly useful and simultaneously so challenging to manage. As it turns back into a gas, it expands with tremendous force. The entire science of cryogenic storage is built around keeping that transition under control.
The implications of liquid oxygen’s boiling point are huge. Every piece of equipment, every safety procedure, and every application is designed with this deep freeze in mind.
To get a clearer picture of how oxygen's properties shift with temperature, here’s a quick comparison.
This table offers a quick-reference look at how oxygen's key physical properties change as it moves from gas to liquid to solid, highlighting the critical temperature thresholds that define its state.
| Property | Gaseous Oxygen (at STP) | Liquid Oxygen (at boiling point) | Solid Oxygen (at melting point) |
|---|---|---|---|
| Temperature | 20°C (68°F) | -183°C (-297°F) | -218.79°C (-361.82°F) |
| State | Gas | Liquid | Solid |
| Density | 1.429 kg/m³ | 1,141 kg/m³ | 1,351 kg/m³ |
| Appearance | Colourless, odourless gas | Pale blue, transparent liquid | Pale blue crystalline solid |
As you can see, the shift from gas to liquid involves a massive increase in density, which is precisely why liquid storage is so efficient. Each state has its own unique characteristics, but for most industrial and medical applications, that liquid state at -183°C is the one we work with.
Keeping a constant, accurate watch on the temperature isn't just good practice—it's essential for safety and efficiency. To do this reliably, you need the right tools for the job. Understanding the various temperature sensor types available is a foundational step for anyone working in cryogenics. Choosing the correct sensor for such a demanding environment can be the difference between a smooth operation and a catastrophic failure.
Taking the air we breathe and turning it into an intensely cold liquid might sound like science fiction, but it's a remarkable feat of engineering that hinges on a few core principles of physics. The process, known as cryogenic air separation, is less about chemistry and more about a carefully controlled dance of pressure and temperature.
It all begins with the ordinary air around us, which is a mix of about 78% nitrogen, 21% oxygen, and trace amounts of other gases.
First things first, the air needs to be cleaned up. It's filtered to remove any dust, moisture, and carbon dioxide. If left in, these impurities would freeze solid at the extreme temperatures we're aiming for, quickly clogging up the delicate machinery.
Next, this purified air is compressed to many times its normal atmospheric pressure, a step that also makes it quite hot. To manage this, the hot, compressed air is then run through a series of heat exchangers, bringing its temperature back down to roughly room temperature.
This is where the real magic happens. The cooled, high-pressure air is suddenly allowed to expand through a valve into a lower-pressure chamber. This rapid expansion forces the air molecules to spread out, causing a dramatic drop in temperature.
This powerful cooling phenomenon is known as the Joule-Thomson effect. By repeating this cycle of compression, cooling, and rapid expansion over and over, the air gets progressively colder until it finally reaches the point where it begins to liquefy.
At this stage, what you have is a liquid mixture of both nitrogen and oxygen. Since each gas has a different boiling point, they can be neatly separated through a process called fractional distillation. Think of it as a highly sophisticated filter that sorts the two liquids based purely on temperature.
The liquid air mixture is fed into a tall distillation column. Nitrogen, with its lower boiling point of -196°C, boils first, turning back into a gas. Being lighter, it rises to the top of the column where it’s collected.
As the nitrogen gas leaves, the remaining liquid becomes more and more concentrated with oxygen. This liquid, now almost pure, sinks to the bottom of the column, where it settles into its stable liquid state at the precise sauerstoff flüssig temperatur of -183°C.
This diagram illustrates just how specific these temperature thresholds are for oxygen's physical states.
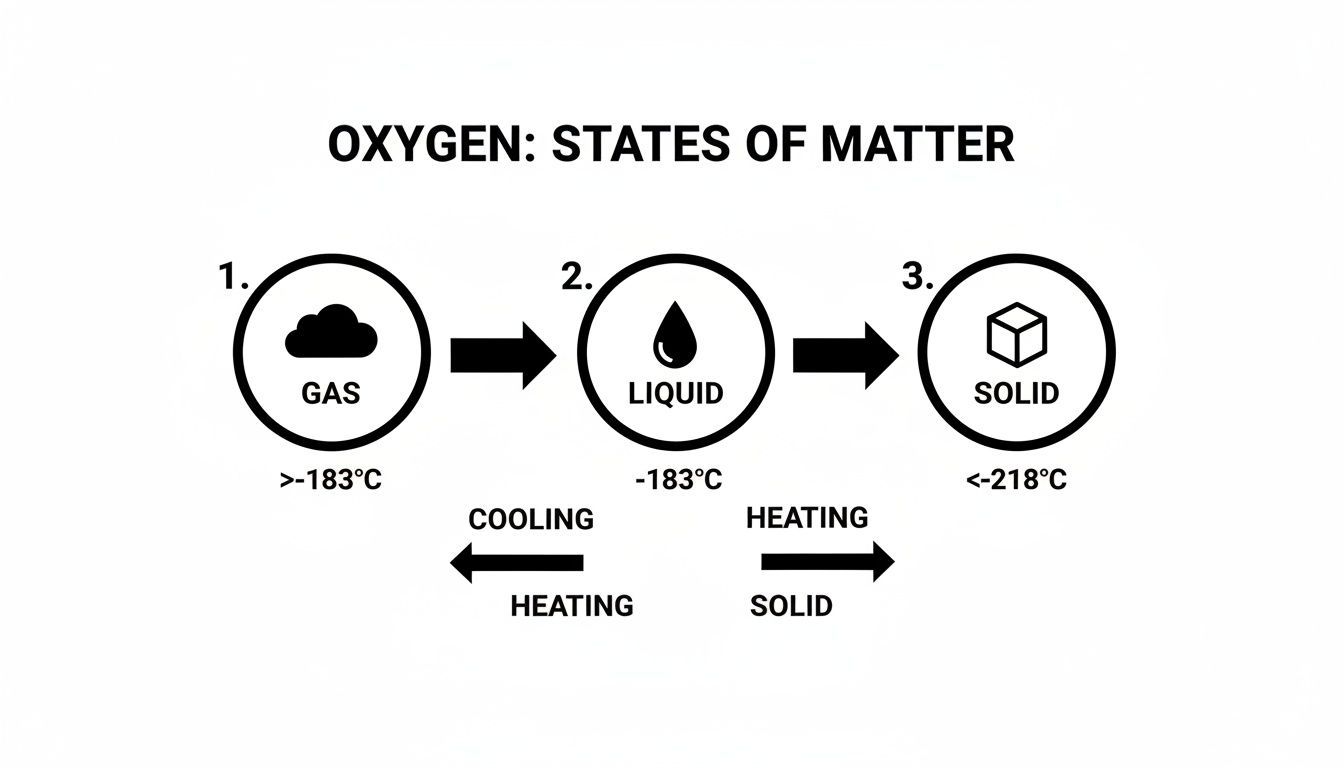
As you can see, the temperature windows for liquid and solid oxygen are incredibly narrow, which really drives home why precise thermal control is so critical in this field. This entire journey, from a simple gas to a potent cryogenic liquid, is a testament to the intricate science behind its production.
To dive deeper into the fundamental nature of this element, you can read our detailed article on the properties of oxygen.

Working with a substance colder than an Antarctic winter demands absolute precision and respect. The extreme sauerstoff flüssig temperatur of -183°C isn't just a number; it introduces a unique set of hazards that call for watertight safety measures. Getting a handle on these risks is the very first step toward creating a truly secure working environment.
The dangers of liquid oxygen go well beyond its intense cold. While cryogenic burns are a major concern, the substance also presents serious asphyxiation and fire risks. Each of these hazards needs its own specific protocols and personal protective equipment (PPE) to be managed properly.
Handling liquid oxygen safely isn't about just one thing—it’s about preparing for its three main dangers. A solid safety plan addresses each one head-on, leaving no room for guesswork or assumptions. These aren't abstract concepts; they are real-world challenges that operators face every single day.
"A single drop of liquid oxygen on unprotected skin is enough to cause a severe cryogenic burn. This isn't just a liquid; it's an extreme thermal hazard that commands the highest level of respect and protection."
Wearing the right PPE is completely non-negotiable when you're around liquid oxygen. Your standard lab or workshop gear is nowhere near adequate and can actually make an accident worse. Every single person handling this substance must be kitted out in specialised cryogenic safety gear.
Your essential PPE checklist should always include:
When dealing with substances like liquid oxygen, strict safety measures are everything. Understanding how to manage these materials effectively is key, and borrowing insights from related fields can be a big help when developing strong protocols for handling hazardous substances.
This forward-thinking approach to safety ensures that your team is not only properly equipped but also mentally prepared for the challenges of working at such a low sauerstoff flüssig temperatur. To dig deeper into the core risks, take a look at our detailed overview of the primary hazards of cryogenic liquids.
 Ever wondered why a standard steel spanner would shatter like glass if you dropped it into liquid oxygen? It's not just about the cold. The extreme sauerstoff flüssig temperatur of -183°C does more than just freeze things; it fundamentally changes how many common materials behave on a molecular level.
Ever wondered why a standard steel spanner would shatter like glass if you dropped it into liquid oxygen? It's not just about the cold. The extreme sauerstoff flüssig temperatur of -183°C does more than just freeze things; it fundamentally changes how many common materials behave on a molecular level.
This dramatic shift is all down to a phenomenon known as the ductile-to-brittle transition. At room temperature, a material like carbon steel is ductile—it can bend, stretch, and absorb impacts without breaking. But expose it to cryogenic temperatures, and its molecular structure locks up tight, losing all its ability to deform.
Think of the molecules in the steel as a flexible, interconnected chain. Under normal conditions, they can slide past one another. When flash-frozen by liquid oxygen, that flexibility vanishes. The chain becomes a rigid, brittle lattice, where even a small shock can cause a catastrophic fracture.
Luckily, not all materials suffer this fate. Certain metals and alloys are the undisputed champions of the cold, specifically chosen for cryogenic service because their internal structure stays tough and reliable, even when plunged into deep-freeze conditions. These materials are the bedrock of safe liquid oxygen handling and storage.
Key materials you'll find in cryogenic work include:
The choice of material isn't just a technical detail; it's the single most important factor in ensuring the structural integrity and safety of any system designed to hold liquid oxygen. Getting it wrong is a recipe for disaster.
To put this into perspective, let's look at a simple guide comparing common materials and their fitness for liquid oxygen service. This really highlights why some are trusted industry standards while others are strictly forbidden. Understanding this is absolutely crucial, especially when it comes to designing or maintaining what we know about cryogenic vessels.
| Material | Compatibility with Liquid Oxygen | Reasoning (e.g., Ductility at -183°C) |
|---|---|---|
| Stainless Steel (304/316) | Excellent | Remains strong and ductile; does not become brittle. |
| Carbon Steel | Unsuitable | Becomes extremely brittle and prone to shattering upon impact. |
| Copper and Bronze | Excellent | Strength increases at low temperatures while retaining ductility. |
| Most Plastics and Rubbers | Unsuitable | Become hard and brittle, leading to cracking and seal failure. |
| Aluminium (5000/6000 Series) | Good | Maintains good strength and does not become brittle. |
As you can see, the difference is stark. Materials that are perfectly reliable for everyday jobs can become dangerously unpredictable at -183°C. That's why relying on proven, cryogenic-rated materials isn't just a best practice—it's a non-negotiable safety requirement.
That incredibly cold sauerstoff flüssig temperatur of -183°C isn't just some abstract number from a physics textbook. It's the very engine that drives some of Germany’s most formidable and forward-thinking industries. When liquid oxygen moves from the lab to the factory floor, it becomes a high-purity workhorse, absolutely vital for steelmaking, chemical production, and advanced environmental tech.
Take steel production, for example. Injecting pure oxygen into a furnace burns off carbon and other impurities far more effectively than just using regular air. This process, which is only practical with the dense supply that liquid oxygen provides, means higher quality steel gets made in less time and with lower energy bills. It's a key ingredient in Germany's world-renowned manufacturing efficiency.
The story is much the same in the chemical sector. Here, liquid oxygen is a fundamental reactant needed to create essential compounds like ethylene oxide and propylene oxide—the building blocks for countless products we use every day, from plastics to pharmaceuticals. The ability to store and transport massive amounts of oxygen in its liquid form gives German chemical plants the steady, high-volume supply they need to operate at scale.
But it’s not all about heavy industry. Liquid oxygen is also a quiet hero in environmental innovation, and its use in wastewater treatment is a perfect example.
By enriching aeration basins with pure oxygen, German municipalities and industrial sites can dramatically speed up the biological treatment process. This leads to cleaner water and more efficient operations, connecting cryogenic science directly to the country's environmental goals.
Germany’s leadership in these fields isn't an accident; it’s built on a rock-solid, efficient supply chain for industrial gases. The nation leads the European industrial oxygen market, thanks in large part to its powerhouse steel and chemical sectors. Innovations like oxygen-fuel burners, now used by over 50% of European glass manufacturers, have boosted energy efficiency while slashing CO₂ emissions.
On top of that, more than 120 wastewater treatment plants—many with significant German involvement—have switched to oxygen-enriched processes, achieving a 15-20% faster treatment time. You can read more about these findings on the industrial oxygen market here. These real-world applications show how mastering the challenges of sauerstoff flüssig temperatur translates directly into economic strength and sustainable progress for German industry.

Step away from the industrial sphere, and you'll find liquid oxygen playing a silent, yet absolutely critical, role in Germany's world-class healthcare system. For any large medical facility, shuffling countless individual gas cylinders is a logistical nightmare. The real game-changer is the incredible density of liquid oxygen, a feat made possible only by keeping it at a precise -183°C sauerstoff flüssig temperatur.
This super-cooled liquid is the central respiratory lifeline for an entire hospital. A single, highly insulated storage vessel can supply a whole medical centre, feeding a complex network of pipes that deliver life-sustaining gas directly to a patient's bedside. It's more than just an efficient system; it's a fundamental piece of infrastructure that guarantees a constant, reliable flow of oxygen when every second counts.
From the operating theatre to the recovery ward, liquid oxygen is the unseen source for countless medical applications that form the backbone of modern patient care. Without a bulk supply, the capacity of hospitals to deliver advanced treatment would be drastically reduced.
The magic happens during the transition from a super-cooled liquid to a breathable gas. Just one litre of liquid oxygen expands into roughly 860 litres of gaseous oxygen. This remarkable expansion ratio is precisely why it's the only practical way to meet the colossal oxygen demands of a modern hospital.
Germany's unwavering commitment to high standards of patient care, coupled with its robust medical infrastructure, fuels a steady and growing demand for medical-grade liquid oxygen. This is plain to see in market trends, where Germany stands as a central pillar of the European liquid oxygen market. While industrial applications still hold the largest share, the demand from the healthcare sector is picking up pace.
This growth is bolstered by public-private partnerships focused on funding and upgrading oxygen infrastructure, especially in secondary hospitals. The sheer efficiency of liquid oxygen—which dramatically cuts down on transport frequency and boil-off losses—makes it a perfect fit for Germany's dense network of medical facilities. You can dive deeper into these market dynamics in the Germany liquid oxygen devices report.
Ultimately, maintaining the correct sauerstoff flüssig temperatur is far more than just a technical detail. It’s a societal necessity that sustains a lifeline for countless patients every single day.
Working with liquid oxygen, or sauerstoff flüssig temperatur, naturally brings up some important questions. Here are straightforward answers to some of the most common queries we get from professionals in the field.
The moment liquid oxygen’s temperature climbs above its boiling point of -183°C, it starts to boil vigorously. This process, called vaporisation, causes the oxygen to rapidly expand back into a gas, increasing its volume by a staggering factor of about 860 to 1. This is why proper venting on storage tanks is non-negotiable—it's the only way to safely manage the immense pressure build-up.
It all comes down to density. Storing oxygen as a liquid is vastly more efficient in terms of space. Think about it: a single, compact cryogenic tank of liquid oxygen holds the same amount as hundreds of bulky, heavy, high-pressure gas cylinders. For any large-scale industrial or medical operation, it's the only practical way to go.
A common misconception is that liquid oxygen is inherently explosive. Oxygen itself doesn't burn, but it acts as a powerful accelerant. An oxygen-rich atmosphere makes other materials incredibly flammable, which is why strict fire prevention is always the number one safety priority.
For state-of-the-art cryogenic solutions designed for the safe storage and transport of industrial gases and biological samples, trust the experts at Cryonos GmbH. Explore our compliant, high-performance vessels and equipment at https://www.cryonos.shop.