No Products in the Cart
Every single property of oxygen—from the way it fuels our bodies to its heavy lifting in industry—stems from one simple fact: it exists as a diatomic molecule, O₂. Its most critical characteristics include its power as a strong oxidising agent, its transformation into a pale blue cryogenic liquid at -183 °C, and its unique magnetic personality.
Before diving into the technical specs, it's crucial to remember we're almost always talking about O₂, two oxygen atoms bonded together. This is the stable form we breathe and store in cylinders. It’s the properties of this molecule, not the single atom, that dictate its real-world behaviour, whether in a hospital ward or a welding workshop.
Thinking about oxygen's phase changes is a lot like thinking about water, just at ridiculously cold temperatures. Just as water goes from ice to liquid to steam, oxygen shifts states too.
This transformation is the key to storing and transporting it efficiently. Liquefaction allows a massive volume of gas to be compressed into a relatively small, manageable container, making the logistics for medical and industrial supply chains possible in the first place.
For professionals who need accurate data at their fingertips, the table below summarises oxygen's most important properties. These values are the foundation for safety protocols, equipment design, and process control in biobanks, labs, and industrial plants. Getting these numbers right is the first step to handling oxygen safely and effectively.
Here's a critical fact to keep in mind: when liquid oxygen evaporates, it expands to 861 times its original volume. This massive expansion is exactly why proper ventilation and pressure relief systems are non-negotiable for cryogenic storage—it’s what prevents catastrophic container failure.
This table serves as a handy reference, breaking down the key data we'll explore in more detail.
This quick reference table summarises the most important properties of oxygen, providing essential data for scientific and industrial applications.
| Property | Value | Significance |
|---|---|---|
| Boiling Point | -183.0 °C (-297.3 °F) | Defines the temperature needed for liquefaction and storage in its cryogenic state. |
| Density (Liquid) | 1,141 kg/m³ (at -183 °C) | Allows enormous quantities to be stored compactly in cryogenic vessels. |
| Density (Gas) | 1.429 kg/m³ (at 0 °C) | Slightly denser than air, meaning it can pool in low-lying, poorly ventilated areas. |
| Solubility in Water | 30.5 mg/L (at 25 °C) | Limited solubility, but just enough to support all aquatic life on Earth. |
| Chemical Formula | O₂ | This diatomic form is the one that matters for virtually all biological and chemical reactions. |
Understanding these fundamental values is essential before moving on to how oxygen behaves in the real world, from its reactivity to its unique magnetic properties.
Beyond its vital role in chemistry and biology, oxygen has a fascinating physical character, one defined by extreme temperatures and some pretty unique molecular traits. The most crucial physical property of oxygen for industrial and medical use is its ability to transform from a gas into a cryogenic liquid. This change is what unlocks immense possibilities for storing and transporting it. It's not just some abstract scientific curiosity; it's the very principle that makes large-scale oxygen supply feasible.
To get your head around this, think about how temperature and pressure are partners. A pressure cooker uses high pressure to raise water's boiling point, cooking food faster. In reverse, a fridge uses a cycle of pressure changes to lower temperatures. Oxygen follows similar rules, but at a much more extreme level. Its behaviour is dictated by critical thresholds that define whether it's a gas, liquid, or solid.
The two most important numbers in oxygen's physical profile are its boiling and melting points. These aren't just figures for a textbook; they are the operational boundaries that dictate every single aspect of handling it, from the design of storage vessels to the personal protective equipment you need to wear.
Keeping temperatures below that -183°C mark is the fundamental challenge of cryogenic logistics. For a deeper look at the specific conditions required, you can learn more about the temperature of liquid oxygen in our detailed article. This constant battle against ambient heat is exactly why specialised cryogenic vessels are so essential.
This infographic neatly sums up the key physical properties that are directly influenced by these temperature thresholds.
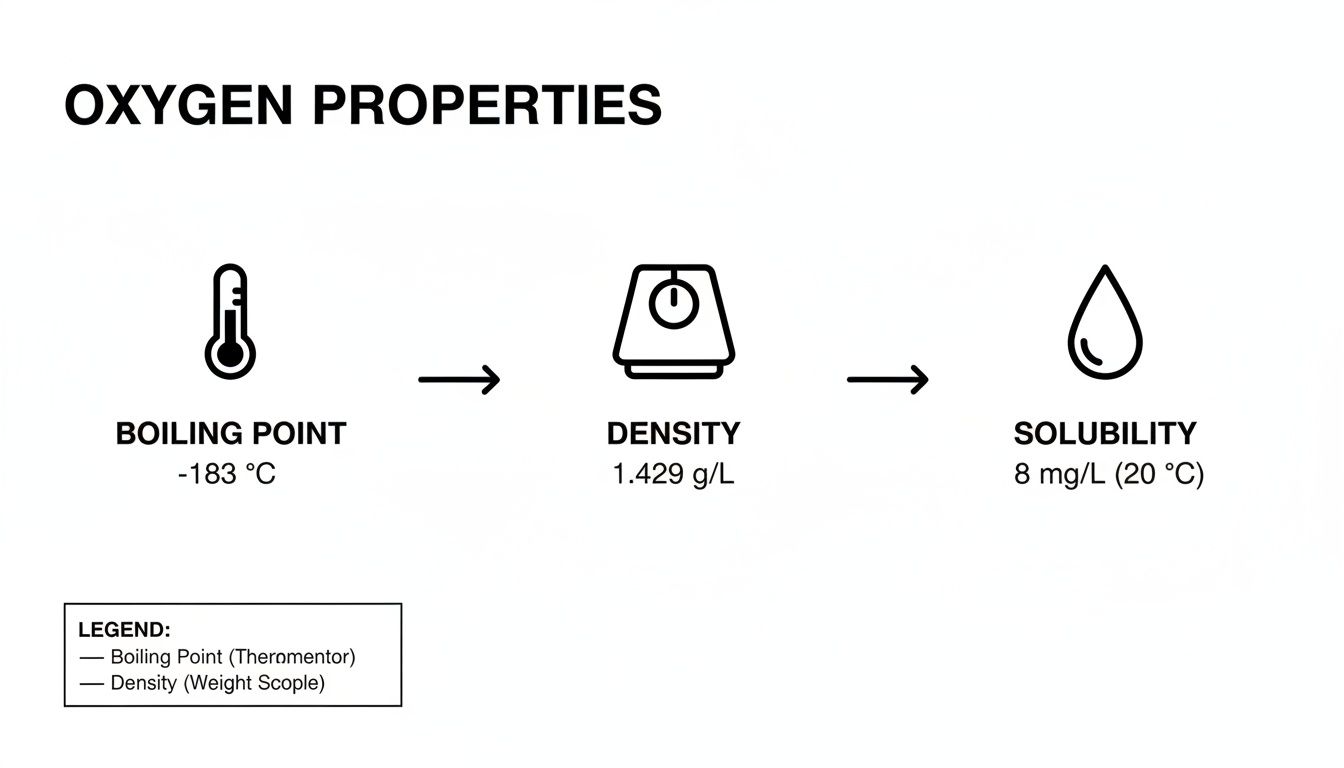
You can see a clear link here: the extremely low boiling point is directly connected to the high density of liquid oxygen and its limited, yet life-sustaining, solubility in water.
One of the most practical physical properties of oxygen is the dramatic change in its density when it becomes a liquid. As a gas at room temperature, it’s diffuse and takes up a huge amount of space. But as liquid oxygen (LOX), it is over 800 times denser.
This incredible densification is a massive advantage. It means a volume of gas that would fill a large room can be squeezed into a compact, manageable cryogenic tank. This is the core principle making bulk oxygen transport and storage both economical and practical for hospitals, biobanks, and industrial facilities. Without liquefaction, the logistics would be almost impossible.
The main reason we produce liquid oxygen isn't to use it as a liquid. It's to take advantage of its high density for efficient storage and transport before it’s turned back into a gas where it's needed.
Perhaps the most surprising physical property of oxygen is its paramagnetism. Unlike most other elements, oxygen is weakly attracted to magnetic fields. This might seem like a minor quirk, but it stems directly from its molecular structure and has some real-world implications.
Imagine each O₂ molecule as a tiny, spinning bar magnet. In its gaseous state, these molecular magnets are all pointing in random directions, so their effect cancels out. But when you cool oxygen into a liquid, they can align with an external magnetic field, creating a noticeable attraction. You can even see this in demonstrations where liquid oxygen is suspended between the poles of a powerful magnet.
This property isn't just a party trick. It plays a role in certain industrial processes and is a consideration in the design of equipment that uses strong magnetic fields, like MRI machines or specific scientific instruments. It’s a perfect example of how a fundamental quantum property can show up in observable, practical ways, adding another layer to oxygen's complex physical identity.
While oxygen's physical properties are fascinating, it's the chemical side of its personality that truly defines its role on our planet. This chemical reactivity is the engine behind countless processes, both natural and industrial, from the slow rusting of a metal gate to the intense, roaring energy of a rocket launch.
It all comes down to one simple, fundamental drive: oxygen’s hunger for electrons.
At its heart, oxygen is a powerful oxidising agent. This is just a technical way of saying it's exceptionally good at stealing electrons from other substances. This process, known as oxidation, isn't just about things going rusty; it’s a core chemical reaction that unlocks and releases energy.
Think of it like a tiny energy transaction. When oxygen gets close to another element, like iron or carbon, it forcefully pulls electrons away. This transfer of electrons breaks the chemical bonds holding the other substance together, unleashing the stored energy as heat and light.
So, why is oxygen so aggressive in its hunt for electrons? The answer lies in its atomic structure. It possesses a very high electronegativity—a measure of an atom's ability to attract and cling to electrons. In the grand league of elements, oxygen is a top contender, second only to fluorine.
This powerful pull makes it reactive with a massive range of other elements and compounds. It loves to form stable chemical bonds, and doing so often releases a significant burst of energy. This is the simple yet profound reason why things burn.
The most dramatic and familiar example of oxygen's reactivity is, of course, combustion—or burning. For a fire to kick off and keep going, you need three things to be present at the same time. You might have heard of it as the fire triangle.
Take away any one of these three components, and the fire goes out. This is the fundamental principle behind nearly all firefighting techniques, whether you're smothering flames with a blanket or dousing them with water to remove heat.
The air around us contains about 21% oxygen, which is more than enough to support normal combustion. But if you start to enrich that oxygen concentration, its reactivity shoots through the roof. Materials that might only smoulder in normal air can become dangerously explosive in a pure oxygen environment.
This is precisely why such strict safety protocols are enforced in hospitals, labs, and industrial sites where concentrated oxygen is used. An oxygen-enriched atmosphere can turn everyday materials into serious fire hazards, where even a tiny spark could trigger a catastrophic fire.
Because oxygen's reactivity is so central to combustion, these principles tie directly into practical safety measures. Understanding general fire risk assessment guidelines is crucial for ensuring safety in any environment where oxygen is stored or handled.
While its reactive nature brings risks, it's also incredibly useful. Across many industries, we've learned to master this powerful property to drive critical processes. Controlling this key eigenschaft von sauerstoff is the secret to both efficiency and safety in these applications.
In every one of these examples, the goal is the same: to leverage oxygen's powerful oxidising nature to release a tremendous amount of energy in a controlled and useful way. From building the backbone of our modern infrastructure to venturing into space, mastering oxygen's chemical reactivity has been fundamental to human progress.
Talking about oxygen's properties is one thing, but putting that knowledge into practice—especially with liquid oxygen (LOX)—is a whole different ball game. LOX isn't just cold; it exists at a temperature that completely redefines what we think of as "freezing." To store and move this cryogenic fluid safely, you need specialised equipment and a healthy respect for the physics that govern its behaviour.
At the centre of all LOX logistics are specialised cryogenic vessels, often called dewars or cryogenic tanks. These are absolutely non-negotiable. Trying to use a standard container would be like trying to carry water in a sieve—it would fail instantly and catastrophically as the LOX boiled off, creating an immense amount of pressure.

The real secret to a high-performance cryogenic vessel is its ability to fight a constant battle against heat. The easiest way to understand the technology is to think of it as a highly advanced thermos flask, built around the core principle of vacuum insulation.
These vessels are built with two walls: an inner one that holds the LOX and an outer one exposed to the environment. The space between them is a near-perfect vacuum. With almost no air molecules in that gap, heat has an incredibly difficult time transferring from the warmer outside world to the frigid liquid inside. This design drastically slows down heat transfer, which in turn reduces how fast the LOX evaporates, or "boils off."
High-quality insulation isn't just a safety feature; it's an economic one. A vessel with superior vacuum integrity and multi-layer insulation will minimise product loss over time, directly translating into lower operational costs for biobanks, hospitals, and industrial users.
This efficiency is what preserves valuable biological samples or ensures a hospital has a consistent, life-saving supply of medical-grade gas.
Another piece of the cryogenic engineering puzzle is materials science. You can't just build these tanks from any old metal. Many common materials, like carbon steel, become terrifyingly brittle at cryogenic temperatures and can shatter like glass on impact.
Instead, engineers must select specific materials that keep their strength and flexibility even when plunged to the extreme cold of -183°C.
Choosing the right material is a fundamental safety decision. It guarantees the structural integrity of the container, preventing failures that could lead to dangerous spills or even explosive ruptures.
Understanding the equipment is only half the battle. Sticking to strict safety protocols is absolutely paramount. The unique eigenschaft von sauerstoff (property of oxygen) in its liquid state creates several distinct hazards that demand careful management and the right personal protective equipment (PPE).
One of the most immediate dangers is pressure build-up. As LOX warms, it expands massively. Every cryogenic vessel must have pressure relief valves to safely vent the gas that naturally boils off. A blocked or faulty vent can turn a storage tank into a potential bomb.
Of course, the extreme cold itself is a massive risk. Even a brief splash of LOX can cause severe cryogenic burns, which are just as damaging as frostbite. Proper PPE is non-negotiable:
Finally, there's the silent but deadly threat of material incompatibility. As a powerful oxidiser, oxygen reacts violently with hydrocarbons like oil and grease. Even a tiny amount of contamination inside a tank or its fittings can lead to a spontaneous, violent fire. That's why all equipment used for oxygen service must be scrupulously cleaned and certified as "oxygen-safe." It's vital to understand the full spectrum of risks, which you can explore in our comprehensive guide to the hazards of cryogenic liquids here: https://cryonos.shop/blogs/cryogenic/hazards-of-cryogenic-liquids.
The growing reliance on medical oxygen, particularly in Germany, really highlights how important these safety standards are. After the COVID-19 pandemic, demand for medical-grade oxygen shot up, pushing the German market to generate revenues of about USD 2,287.1 million in 2022. Projections show it climbing towards USD 4,193.2 million by 2030. This growth has put an even stronger focus on certified storage and transport equipment that meets strict medical and transport regulations, reinforcing the need for safe, reliable cryogenic solutions. You can find more insights about the growth of Germany's industrial gas market on ibisworld.com.
You might think oxygen is just oxygen, but that's a dangerous oversimplification. The oxygen used in a hospital is fundamentally different from the oxygen used to cut steel, and understanding why is critical. This isn't just a minor technical detail; it’s the cornerstone of performance, safety, and regulatory compliance.
Every application has its own specific purity requirements. Using the wrong grade isn’t just a mistake—it can lead to catastrophic equipment failures, completely ruined experiments, or even serious harm to patients. The real difference lies in the acceptable levels of impurities. Even minuscule traces of other gases, moisture, or particulates can have a massive impact, which is why oxygen is carefully categorised into distinct purity grades.

The most important distinction to grasp is between medical and industrial oxygen. Medical-grade oxygen is treated as a prescription drug, and for good reason—it's subject to incredibly strict regulations to guarantee patient safety.
Medical-Grade Oxygen: This must have a minimum purity of 99.0%. But more importantly, it must be completely free from specific contaminants like carbon monoxide, carbon dioxide, and other trace gases that could be harmful to breathe in. The entire supply chain, from the production plant to the cylinder arriving at a patient's bedside, is rigorously validated to prevent contamination.
Industrial-Grade Oxygen: Here, purity levels can vary quite a bit, from around 99.5% for general welding up to ultra-high purity grades for niche applications. The critical difference is that the chain of custody isn't validated for human use. This means cylinders could easily contain contaminants leftover from previous industrial processes.
Using industrial oxygen for medical purposes is extremely dangerous and illegal. The risk of introducing harmful contaminants directly into a patient's respiratory system is simply unacceptable. For any medical application, certified equipment and a fully validated supply chain are absolutely non-negotiable.
Beyond the basic medical and industrial divide, there's a whole other world of high-purity oxygen designed for highly sensitive scientific and manufacturing processes. In fields like semiconductor manufacturing or advanced lab analysis, even the tiniest impurity can ruin a multi-million-dollar batch of microchips or skew vital research data.
These applications often demand "five-nines" (99.999%) or even "six-nines" (99.9999%) purity. Achieving and maintaining this level requires specialised production methods, dedicated analytical gear, and certified storage vessels built to prevent any hint of contamination. Different uses demand specific purity levels, such as when using devices like oxygen concentrators. To get a better feel for how these work, you can learn more about oxygen concentrators and their output purity.
The pricing for these different grades reflects just how complex they are to produce and handle. The market dynamics in Germany show this trend clearly; with the domestic oxygen market projected to grow from USD 2,287.1 million in 2022 to USD 4,193.2 million by 2030, the demand for certified, high-purity grades is on the rise. This growth directly impacts how labs and hospitals procure their supplies, making smart storage and supply strategies essential for managing costs.
Ultimately, picking the right grade means balancing cost, purity requirements, and logistics. Making the right call between buying or renting gas cylinders is a huge part of this strategy, which you can dive into deeper in our guide to gas cylinder procurement.
Even when you feel like you have a good handle on oxygen's properties, questions still pop up in the day-to-day. This section is all about tackling some of the most common queries we hear from professionals working with this essential element. The goal is to give you direct, clear answers to clear up any doubts, reinforce critical safety points, and offer advice you can use right away.
It catches a lot of people by surprise, but liquid oxygen actually has a beautiful, pale blue colour. This isn't a sign of an impurity; it's a natural physical property tied directly to its molecular structure. It all comes down to how O₂ molecules interact with light when they're packed so tightly together in their liquid state.
In that dense, cold environment, pairs of O₂ molecules can form temporary, loose bonds. These fleeting pairings are just right for absorbing light from the red end of the visible spectrum. When you take red light out of the full spectrum of white light, what's left for our eyes to see is its complementary colour—blue. It’s a subtle effect, but a fantastic visual demonstration of an eigenschaft von sauerstoff (property of oxygen).
This is a critical question, and the answer really has two parts. While oxygen’s ability to aggressively fuel a fire is a massive hazard, the most immediate danger to you is the extreme cold. At a bone-chilling -183°C (-297°F), even a tiny splash of liquid oxygen on your skin will cause severe cryogenic burns. These are just like severe frostbite and can cause permanent tissue damage in an instant.
The second huge risk is over-pressurisation. This one is a silent but incredibly powerful danger. As liquid oxygen warms up, it boils and expands to 861 times its liquid volume. If that expansion happens inside a sealed container with no vent, the pressure can build to a catastrophic level, causing the vessel to rupture with the force of an explosion. This is why proper personal protective equipment (PPE) and correctly functioning, vented equipment are absolutely non-negotiable.
When thinking about risks, here’s the order of operations: First, protect yourself from the cold with the right PPE. Second, make sure your equipment is properly vented to handle the pressure. Both of these hazards can cause severe injury or damage if you don't give them the respect they demand.
Let’s be perfectly clear: the answer is an absolute and unwavering no. Using industrial-grade equipment for medical oxygen is incredibly dangerous, illegal in most places, and flies in the face of strict regulatory standards designed to keep patients safe. The two systems are built to completely different specifications, especially when it comes to cleanliness and material compatibility.
Here’s a quick rundown of why this separation is so vital:
Always, always ensure that every single component in a medical gas delivery system—from the storage tank right down to the final delivery point—is specifically certified for medical oxygen service. There are no shortcuts here.
Cutting down on oxygen loss, often called boil-off, is key for managing costs and keeping your operations efficient. This loss happens when heat inevitably leaks into the cryogenic vessel, causing the liquid inside to warm up and evaporate. Tackling this problem really comes down to two things: the quality of your storage vessel and your handling procedures.
High-performance cryogenic tanks are built with sophisticated insulation systems. They usually have a multi-layer super-insulation blanket tucked into a high-vacuum space between the tank's inner and outer walls. This design is incredibly effective at stopping heat transfer in its tracks.
To cut down on boil-off even further, follow these best practices:
Ultimately, investing in a high-quality, well-maintained vessel from a trusted supplier will dramatically reduce your long-term product loss and operational costs. It ensures you get the most out of every litre you buy.
For state-of-the-art cryogenic solutions designed for maximum safety, efficiency, and minimal product loss, trust Cryonos GmbH. We provide turn-key systems for the storage, transport, and handling of biological samples and industrial gases, backed by decades of expertise. Explore our range of medically licensed and ADR-compliant cryogenic vessels at https://www.cryonos.shop.